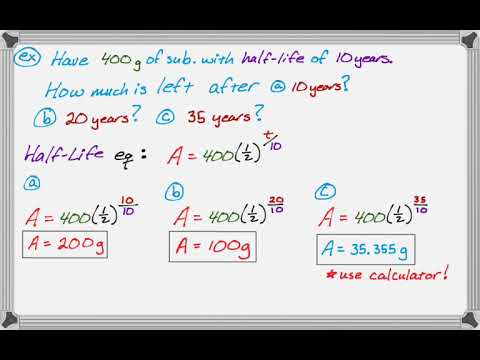half life formula chemistry
The unit of half-life equation for zero order reaction is second 2. Using the half-life equation derived from the concentration-time equation as shown in example 1 we can solve for the initial concentration of reactant.

Half Life Of A First Order Reaction Video Khan Academy
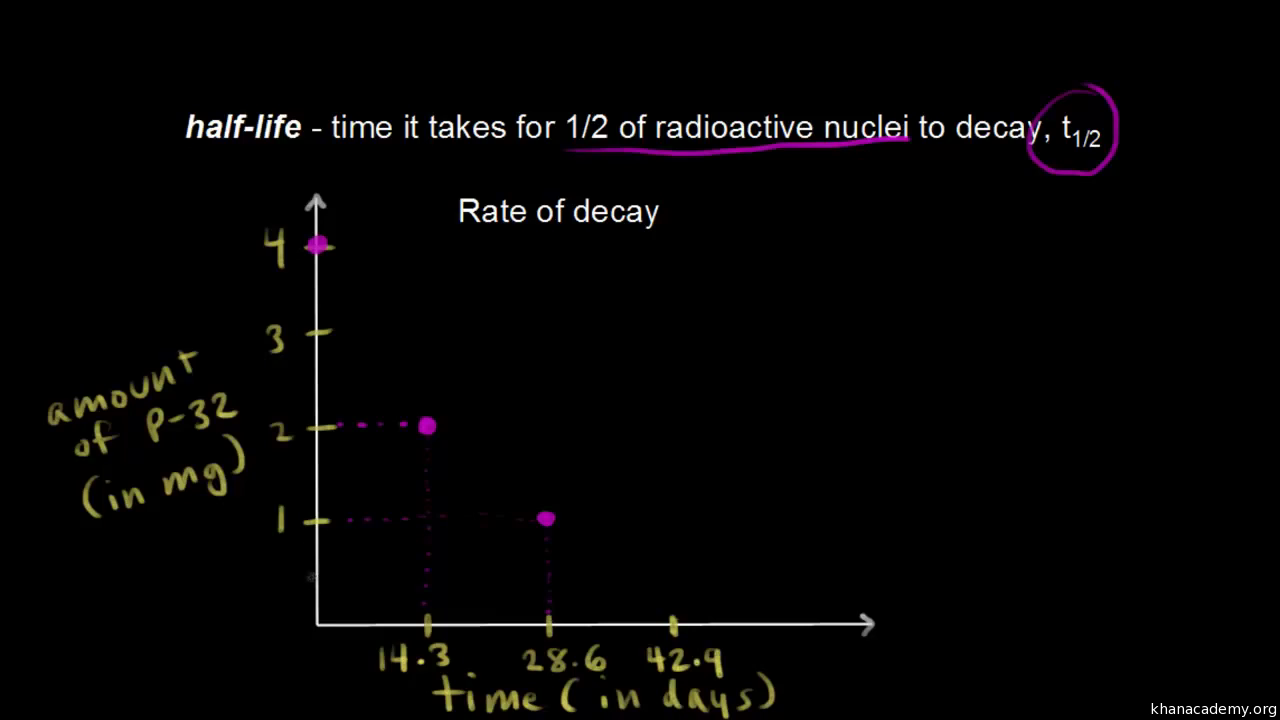
Chemical half-life is the amount of time needed for half of a radioactive substance to decay.
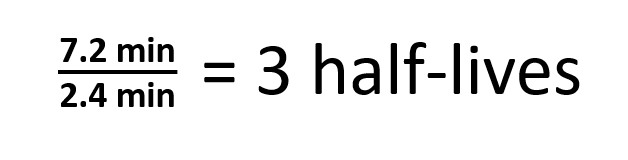
. Half-life can be calculated by the given formula. Department of Chemistry Boston University Boston MA 02215 Using coin flips to count people Everyone in of a group of 202 people is initially standing. It is important to note that the formula for the half-life of a reaction varies with the order of the reaction.
N t N 0 05 t n. Here λ is called the disintegration or decay constant. The half-life of a reaction t 1 2 is the time required for an initial reactant concentration A 0 to decrease by one-half.
The half-life has importance for the quantification. Therefore A t 1 2 A 0 at t 1 2 The half-life. We utilize the equation that relate amount remaining initial mass and number of half-livesn.
Half life formula of nth order reaction is t 1 2 2 n 1 1 A 0 n 1 n 1 k Problem practice If the half life period for a first order reaction in A is 2 minutes How long will it take. This formula is used where the rate of decay of the substance at any instant is. What is the formula for calculating half-life.
Each person flips a coin and. Nt No e -λ t12 Why do we calculate half-life. What is the half-life formula for chemistry.
We can determine that the order of a decay reaction is 1 by looking at the. So the value of t can be found or determined using half life equation. The half-life formula for various reactions is given below.
The formula for the half-life is obtained by dividing 0693 by the constant λ. N 0 is the. N t is the remaining quantity of a substance after time t has elapsed.
Hence the formula to calculate the half-life of a substance is. T ½ 1 k A o Top Determining a Half Life To determine a half life t ½ the time required for the initial concentration of a reactant to be reduced to one-half its initial value we need to know. For a zero-order reaction the mathematical expression that can be.
Half-life formula and unit for first order reaction. Half-Life is the required time for a sample of radioactive material to reach half the mass of the original sample. C-14 Half-Life 5730 Years.
The mathematical expression that can be employed to determine the half-life for a zero-order reaction is t12 R 02k For the first. N t 1 2 n X N o 1 2 4 X 50 3125 g. The half-life formula used to calculate zero order reaction is t₁₂ A₀2k.
This concept is used in carbon dating. The Half-Life formula is used to find the time a substance will decay to half of its initial value. Half life chemistry problems nuclear radioactive decay calculations practice examples you calculator tips for concept by brightstorm definition formula calculation 5 ways.
T_ 12 frac 1.

Using The Half Life Formula For More Complex Problems Science Chemistry Nuclear Chemistry Showme

Radioactive Decay Formula Radioactive Half Life 0 693 Radioactive Decay Constant Physics Topics Science Themes Physics

Half Life Calculator Radioactive Decay Calculator
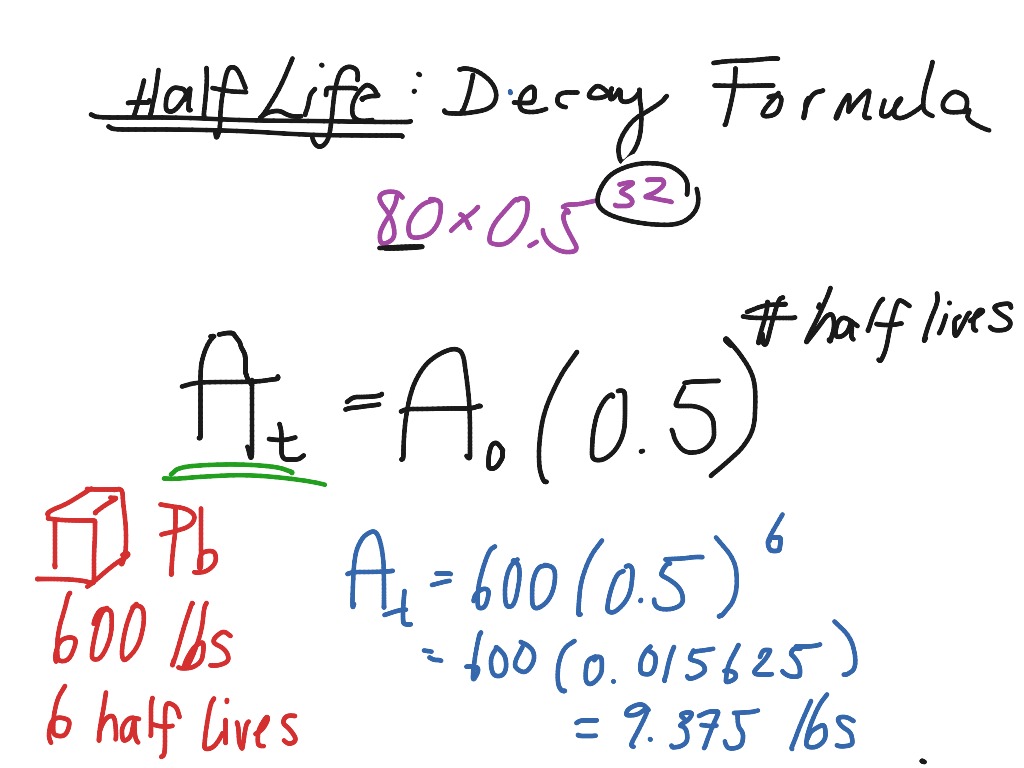
Radioactive Decay Formula Science Chemistry Nuclear Chemistry Showme
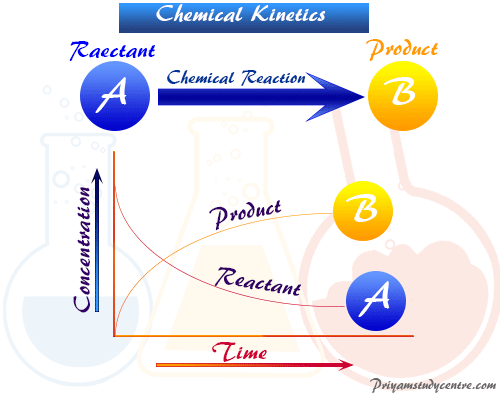
Chemical Kinetics Half Life Definition Formula Equations
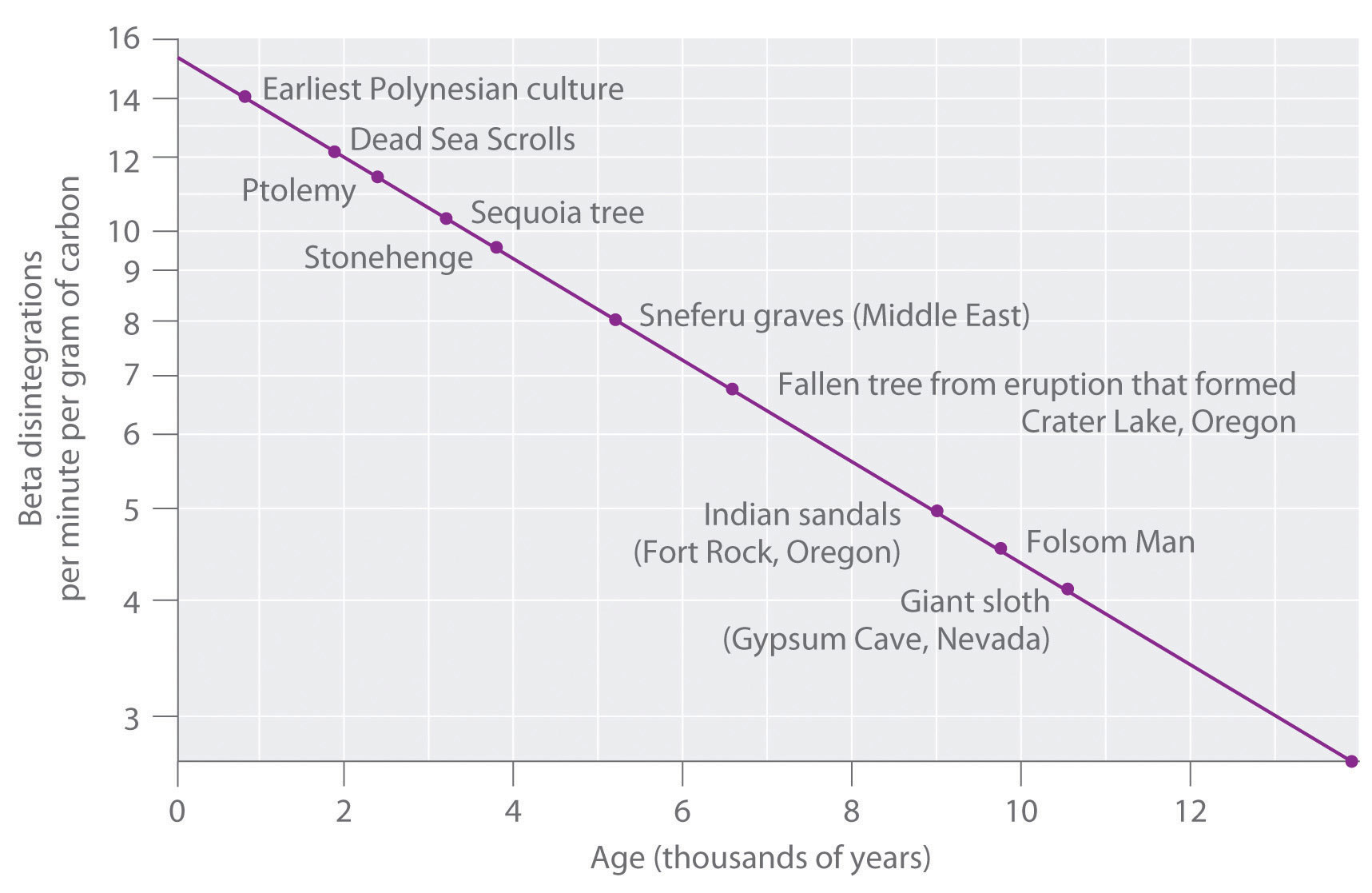
Half Lives And Radioactive Decay Kinetics Chemistry Libretexts

5 Ways To Calculate Half Life Wikihow

Half Life Chemistry Problems Nuclear Radioactive Decay Calculations Practice Examples Youtube
Half Life Introductory Chemistry
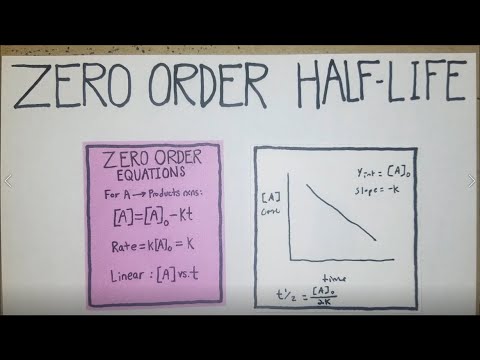
How To Calculate Half Life For Zero Order Reactions Youtube

Half Life Chemistry Problems Nuclear Radioactive Decay Calculations Practice Examples Youtube

5 Ways To Calculate Half Life Wikihow

Rate Equation And Order Of Reaction
Radioactivity Half Life Lessons Blendspace

Ap Chemistry Q3b1 My Ap Chemistry Blog
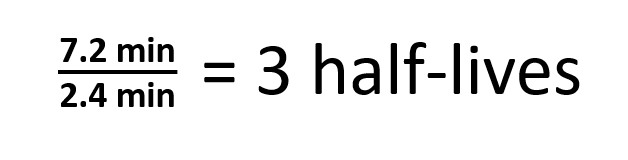
Ch103 Chapter 3 Radioactivity And Nuclear Chemistry Chemistry